Oral contraceptive pill (risk factors for arterial disease)
Last reviewed dd mmm yyyy. Last edited dd mmm yyyy
- a combined pill containing a low dose of oestrogen (i.e. 30 mcg) is indicated in women who have migraines without auras - this is because the risk of stroke is greater the higher the dose of oestrogen in the combined pill
- a DTB review (1) suggested that low-dose pill can also be given to women who have migraines without auras but have one additional risk factor for stroke - however the review emphasises that these patients must be followed up carefully
- prescribing decisions, with respect to combined hormonal contraception, are made based on the UKMEC criteria below which define migraine with an aura as an absolute contraindication to combined hormonal contraception - see below for further details and guidance with respect to different migraine scenarios
UKMEC Criteria state:
Cardiovascular factors (including migraine) and UKMEC categories (3):
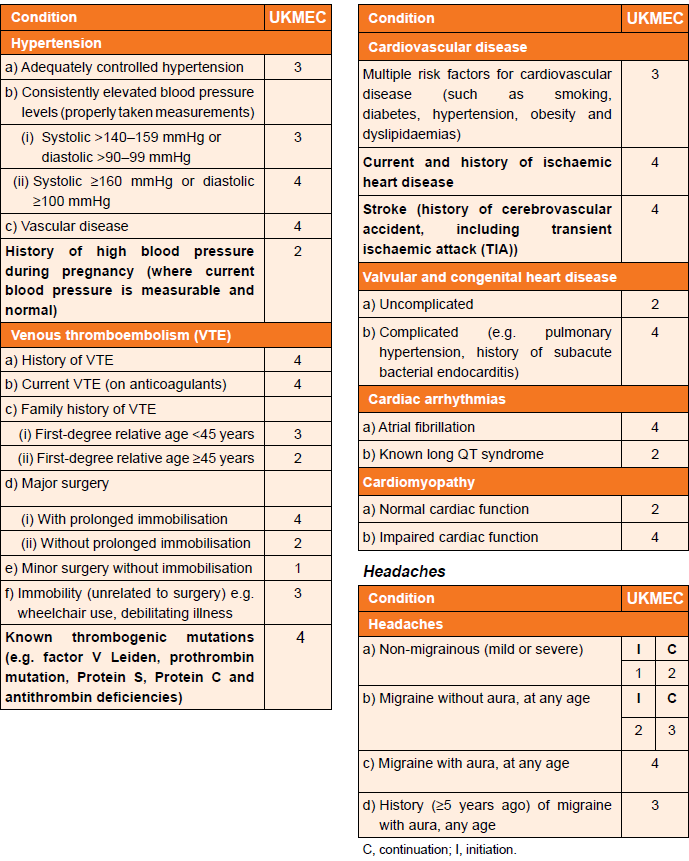
In consideration of UKMEC criteria and combined hormonal contraception (CHC):

Check the summary of product characteristics before prescribing any combined oral contraceptive pill.
Reference:
- (1) Drug and Therapeutics Bulletin (2000), 38 (1), 1-4.
- (2) BNF 7.3
- (3) FSRH Clinical Guideline: Combined Hormonal Contraception (January 2019, Amended July 2019)
Related pages
Create an account to add page annotations
Add information to this page that would be handy to have on hand during a consultation, such as a web address or phone number. This information will always be displayed when you visit this page