Hormone replacement therapy in the management of urogenital symptoms
Last reviewed dd mmm yyyy. Last edited dd mmm yyyy
Oestrogen is used for the management of urogenital symptoms (e.g. vaginal dryness, dyspareunia as a result of vaginal dryness, recurrent urinary tract infection, and urinary frequency and urgency). Alternatives for management are:
- low-dose vaginal oestrogen such as oestriol (cream, pessary or gel) and/or systemic (oral or transdermal)
- NICE state that with respect to the management of urogenital atrophy (1)
- offer vaginal oestrogen to women with urogenital atrophy (including those on systemic HRT) and continue treatment for as long as needed to relieve symptoms
- consider vaginal oestrogen for women with urogenital atrophy in whom systemic HRT is contraindicated, after seeking advice from a healthcare professional with expertise in menopause
- if vaginal oestrogen does not relieve symptoms of urogenital atrophy, consider increasing the dose after seeking advice from a healthcare professional with expertise in menopause
- explain to women with urogenital atrophy that:
- symptoms often come back when treatment is stopped
- adverse effects from vaginal oestrogen are very rare
- they should report unscheduled vaginal bleeding to their GP
- advise women with vaginal dryness that moisturisers and lubricants can be used alone or in addition to vaginal oestrogen
- do not offer routine monitoring of endometrial thickness during treatment for urogenital atrophy
Notes:
- improvement may take several months, and symptoms may recur if treatment is stopped
- long-term treatment is often required. Low-dose vaginal oestrogen may be preferred if the woman does not wish to take systemic HRT or cannot tolerate systemic HRT
- endometrial effects should not be incurred, and a progestogen is not needed with such low dose preparations
- advise women using topical oestrogen therapy to contact their doctor if they experience any vaginal bleeding
- treatment should be reviewed at least annually - " systemic effects of oestrogen are minimised by using the lowest effective dose to control symptoms; the dose may be increased on the advice of a healthcare professional with expertise in menopause if there is inadequate symptom control. Treatment is continued for as long as needed to relieve symptoms and reviewed initially at 3 months, then at least annually" (2)
- if there is no symptomatic improvement with hormonal treatment, then another underlying cause of for the symptoms should be considered (eg, dermatitis, vulvodynia).
Note that these products may damage latex condoms and diaphragms
Topical oestrogens - options include (2):
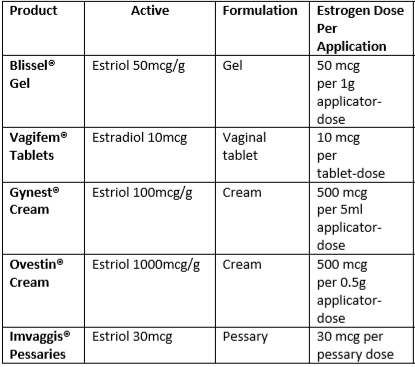
Initiating and monitoring treatment for topical oestrogens (3)
- use the lowest effective dose to minimise systemic absorption - eg, pessaries or creams or gels daily for the first two weeks and then reducing to twice weekly
- it is common to have more vaginal discharge with pessaries and creams, which may be an advantageous side-effect in sexually active women
- topical vaginal oestrogen preparations reverse urogenital atrophic changes and may relieve associated urinary symptoms
- a review from the Collaborative Group on Hormonal Factors in Breast Cancer (4) “...There appeared to be little risk, however, from topical vaginal oestrogen preparations, which limit systemic exposure.”
- use of vaginal oestrogen after diagnosis of breast cancer
- a cohort study of 49,237 females with breast cancer, found that there was no evidence of an increase in early breast cancer-specific mortality with use of vaginal estrogen therapy compared with no hormone replacement therapy use after breast cancer diagnosis (5)
- maximum benefit with these products is usually achieved after around 1–3 months but it can take up to 1 year in some women. Treatment with topical oestrogen should be continued for as long as needed to relieve symptoms as symptoms will often return after treatment is stopped (1) - since the systemic absorption of oestrogen from recommended doses of topical oestrogens is very small (i.e. approximately 1 year's supply of topical therapy contains the same dose as taking a single tablet of oral hormone replacement therapy [HRT]), it is unlikely to be associated with the adverse effects reported with the use of systemic HRT
Contributor/Reviewer:
- Dr Louise Newson,GP and Menopause Specialist (July 14th 2020)

- for further information about HRT and the menopause then see the links below:
All NICE guidance should be viewed in close conjunction with the particular prescribing information (Summary of Product Characteristics) of individual medicinal products cited.
Reference:
- (1) NICE (November 2015). Menopause: diagnosis and management
- (2) British National Formulary (BNF) (November 5th 2023)
- (3) Al-Baghdadi O, Ewies AA; Topical estrogen therapy in the management of postmenopausal vaginal atrophy: an up-to-date overview. Climacteric. 2009 Apr;12(2):91-105
- (4) Collaborative Group on Hormonal Factors in Breast Cancer.Type and Timing of Menopausal Hormone Therapy and Breast Cancer Risk: Individual Participant Meta-Analysis of the Worldwide Epidemiological Evidence.Lancet. 2019 Sep 28;394(10204):1159-1168.
- (5) McVicker L, Labeit AM, Coupland CAC, et al. Vaginal Estrogen Therapy Use and Survival in Females With Breast Cancer. JAMA Oncol. Published online November 02, 2023. doi:10.1001/jamaoncol.2023.4508
Related pages
Create an account to add page annotations
Add information to this page that would be handy to have on hand during a consultation, such as a web address or phone number. This information will always be displayed when you visit this page